Happy April Fools Reef Builders. No, this isn’t another instance of Peanut Butter curing brown jelly disease. Instead, I decided to do something different this year. Many people may not click on this article because they’ll likely see the goofy picture and tagline of dosing liquid gold and assume this is another prank article. It could not be further from that.
Last year, I tried to produce an article that displayed blatant misinformation in a convincing way. This year, I decided to flip that premise on its head and produce an entirely factual article portrayed with a zany cover (hoping that people never even click on the article). To those of you who are here – welcome to another scientific info dump, the likes of which I have not produced since last summer’s DOC article – enjoy.
Setting The Scene
Late last year during some of my first months living in Colorado I went north to Fort Collins to visit the local fish stores. I had no idea that Underwater Wonders would end up being an SPS mecca with some of the best quality of pieces I had ever seen. Upon meeting Seth, Garret, and Cole I knew they were likely doing something different.
I had never before seen Acropora with such vibrant colors. One little secret they revealed to me is that they were dosing a product called AquaVitro Synthesis. Synthesis is a full spectrum nitrogen supplement product designed for freshwater planted tanks. It contains ammonia, nitrate and… urea. I can’t remember for sure but I believe they said they originally picked up this trick from Top Shelf Aquatics.
I spoke with both Seth from Underwater Wonders and Kevin Burda from Top Shelf Aquatics and they confirmed they both still utilize synthesis on their systems at the time of writing this article.
Knowing that two of the best Acropora farms in the country were both doing the same thing and both producing sticks with coloration in the top 2.5+ percentile got my head turning. Immediately, I dove down the rabbit hole.

I went to a local shop close to the studio and bought all of their synthesis. At the time, I was using ammonium bicarbonate on the systems here. I decided to bite the bullet for science and swap out the ammonia for synthesis. I decreased the dosage by around 20% and then slowly ramped it back up to the same dosage I had my ammonia set to after several weeks of seeing no issues.
Anecdotally, I saw very good results. Pieces started to darken, smoothies started to have polyp extension almost 24/7 etc etc etc. To be clear I can’t credit all of this purely to switching to synthesis as I made a few other changes to the system I was testing it on such as turning off the skimmer.
After sitting on the urea question for several months I continued to research. As of now I don’t believe that utilizing synthesis is the best solution for nitrogen supplementation on reef tanks. I also don’t believe purely ammonia or nitrate addition is either.
In the remainder of this article I will break down the current literature base when it comes to nitrogen and coral and describe what I believe may be the best path forward with the available data.
Dosing Nitrate Is Bad?
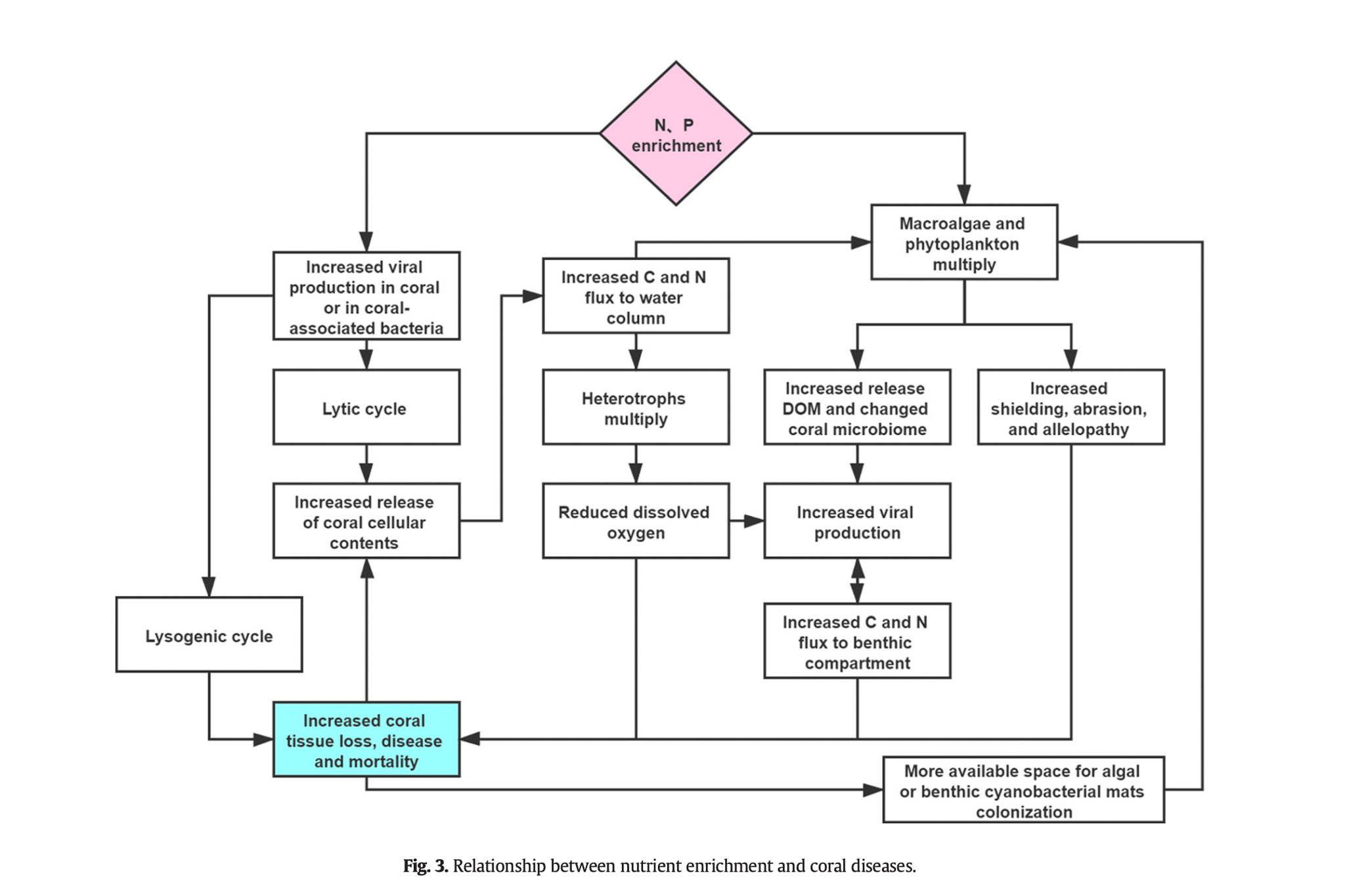
To keep things as short as possible the existing consensus in scientific literature at this time is that exposing corals to excess nitrates is harmful to them. The definition of excess is something which differs for every species and even differs for the same species depending on different microhabitats. Nonetheless the data is clear – excess nitrogen and in particular nitrates harm corals in a multitude of ways which I will list below.
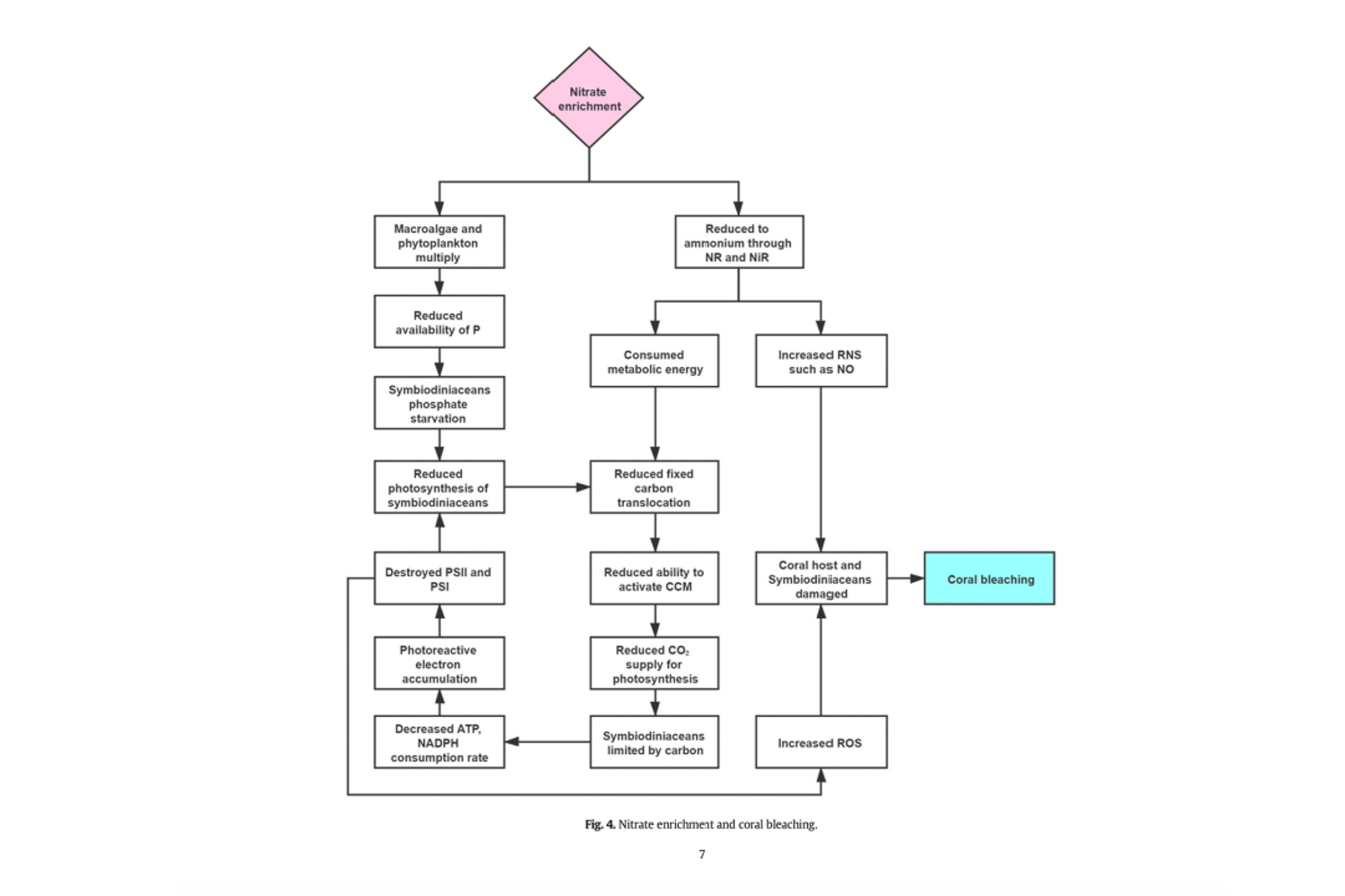
When excess nitrogen was measured on reefs it was found to lead to an imbalance in nitrogen and phosphorus which damaged the corals photosystems and hammered the ability for zooxanthella to give the coral carbon which led to corals starving. It also caused an increase in nitrogen based free radicals.
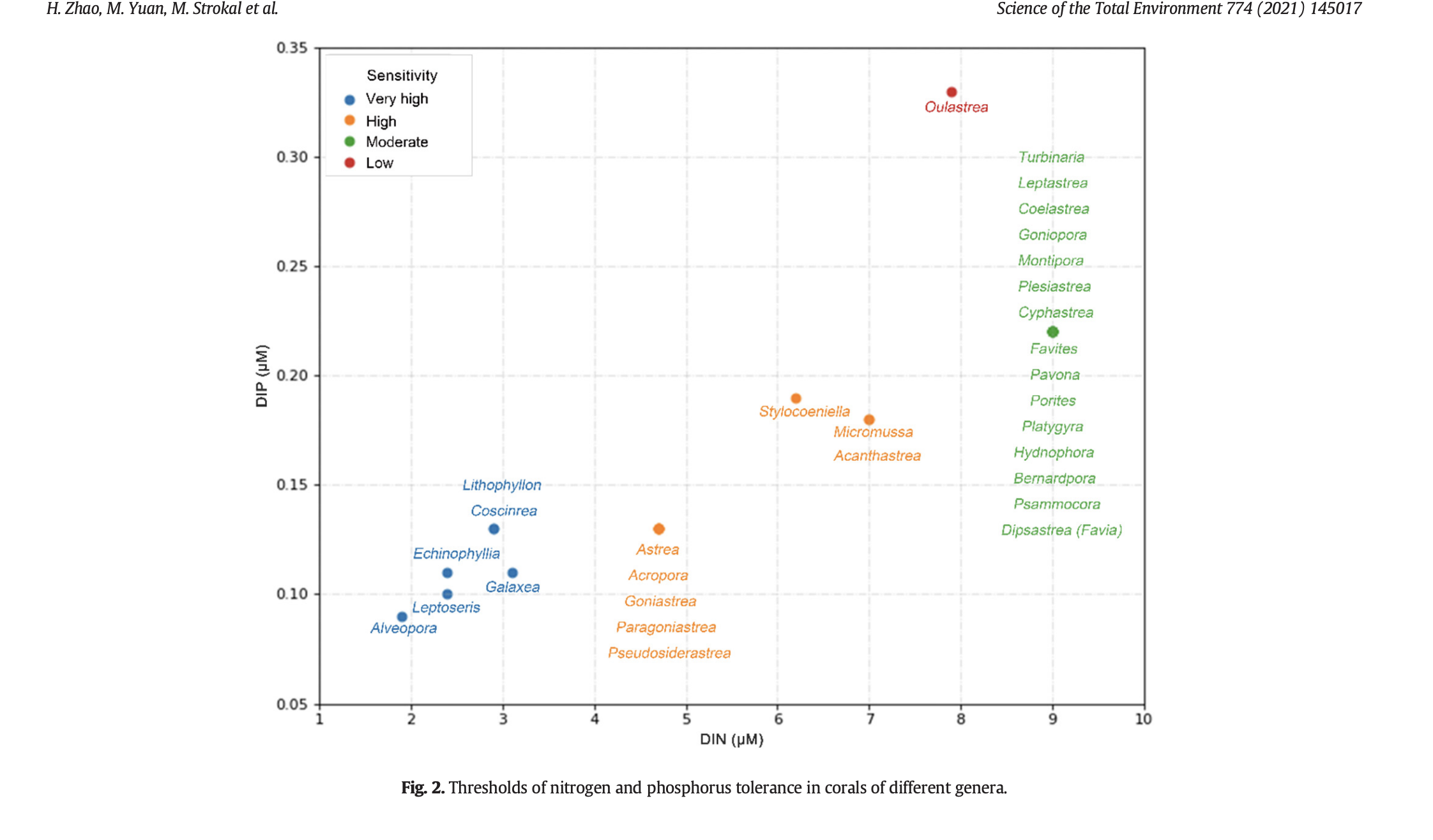
Sensitivity of different genera to inorganic nutrients (maybe this is why some are more hardy in captivity?) [3]
Excess nitrogen can also increase the growth of competing algae, which feeds into the DDAM model and any associated issues with DOC. Environments in the ocean with high nitrogen and heat stress lead to an increased risk of bleaching. In areas with low heat stress, high nitrogen increased the severity of bleaching. [1]
While the initial study looked at nitrogen levels in general, subsequent studies analyzed the effects of nitrate addition in particular (in comparison to the addition of urea) on wild colonies for several years.
It was found that nitrate addition increased the likelihood that corals would bleach but did not affect how severe the bleaching event was. Even though nitrate was not correlated to bleaching severity, it was heavily correlated with colony mortality in both Acropora and Pocillopora colonies. The study also posited that colony morality was due to zooxanthella being exposed to high levels of nitrate and being less efficient at returning carbon to the coral; thus, they slowly starved over time. [2]

Bleach severity correlated to nitrogen addition and heat stress [2]
An experiment conducted by Dr. Rebecca Vega Thurber and co in 2014 added commercial fertilizer that had a balanced mix of nitrate and ammonium, phosphorous, and micronutrients to coral frags in the wild. This created a localized “eutrophic” or high-nutrient environment around the corals. While this study did not differentiate between nitrate and ammonia.
“Human-related sources of eutrophication are usually relatively high in nitrate (Shantz and Burkepile 2014), which suggests that the presence of nitrate outweighs potential benefits of ammonium with regard to causation of diseases. The results obtained from enrichment experiments with nitrate-rich fertilizers (Bruno et al. 2003; Vega Thurber et al. 2014; Zaneveld et al. 2016) support this view.” [3]
The addition of fertilizers increased disease by 170% in Siderastrea siderae. Typical loss due to nutrient exposure is causing Siderastrea to die off at a 1% rate per year. In the nutrient exposure experiments, the population died off at 3% per year. After the nutrient addition experiment ended, the instance of disease returned to normal, adding further proof that the induction of a eutrophic environment is likely a key factor in the development of coral disease. The Vega Thurber paper theorized that excess inorganic nutrients may shift the microbiome to a pathogenic state or reduce corals’ immune system, making them more susceptible to infection.

Coral disease correlated to nutrient enrichment over time [5]
Another interesting study revealed that when corals were exposed to high inorganic nutrients, parrotfish bites (which can typically be beneficial on wild reefs as they aid in new growth, growth management, etc) led to an increased rate of corals getting infected with pathogenic strains of bacteria. This helps support the notion that high inorganic nutrients are likely correlated with disease through some mediator of external stress. [3]
A recent paper published in March of this year also tracked the microbiome of corals exposed to both nitrate and excess heat. It found that corals exposed to both co-stressors had a correlative increase in the Enterobacteriaceae and Vibrionaceae families. Both of these have been shown to be involved with coral disease susceptibility (especially Vibrio). [4]
While some of these studies are in the context of heat + high nitrogen. A review paper tied in high inorganic nutrient values in with the DDAM model and DOC and suggested that elevated DOC may also be a trigger for such stress events.
“Furthermore, Wooldridge [40] proposed a mechanism by which enhanced retention of photosynthates by Symbiodinium due to excess nitrogen availability may cause CO2 limitation, ultimately resulting in coral bleaching. Therefore, environmental stressors such as heat stress and elevated DOC concentrations may potentially lower the threshold at which disruption of the coral–algae symbiosis occurs by increasing coral-associated nitrogen fixation rates.” [6]
“Breakdown of the coral–algae symbiosis and potential subsequent demise of the coral host may, at the same time, trigger a positive feedback loop of coral degradation (Figure 2), which may act on different scales from colony to ecosystem level (Figure 3). Epilithic algae communities on freshly killed corals show increased nitrogen fixation rates and also release DOC-rich exudates 86, 87. These exudates may stimulate microbial activity (including nitrogen fixation) in nearby corals. Thus, algae overgrowth on the dead coral skeleton may stimulate further bleaching and eventually cause mortality in adjacent corals.” [6]
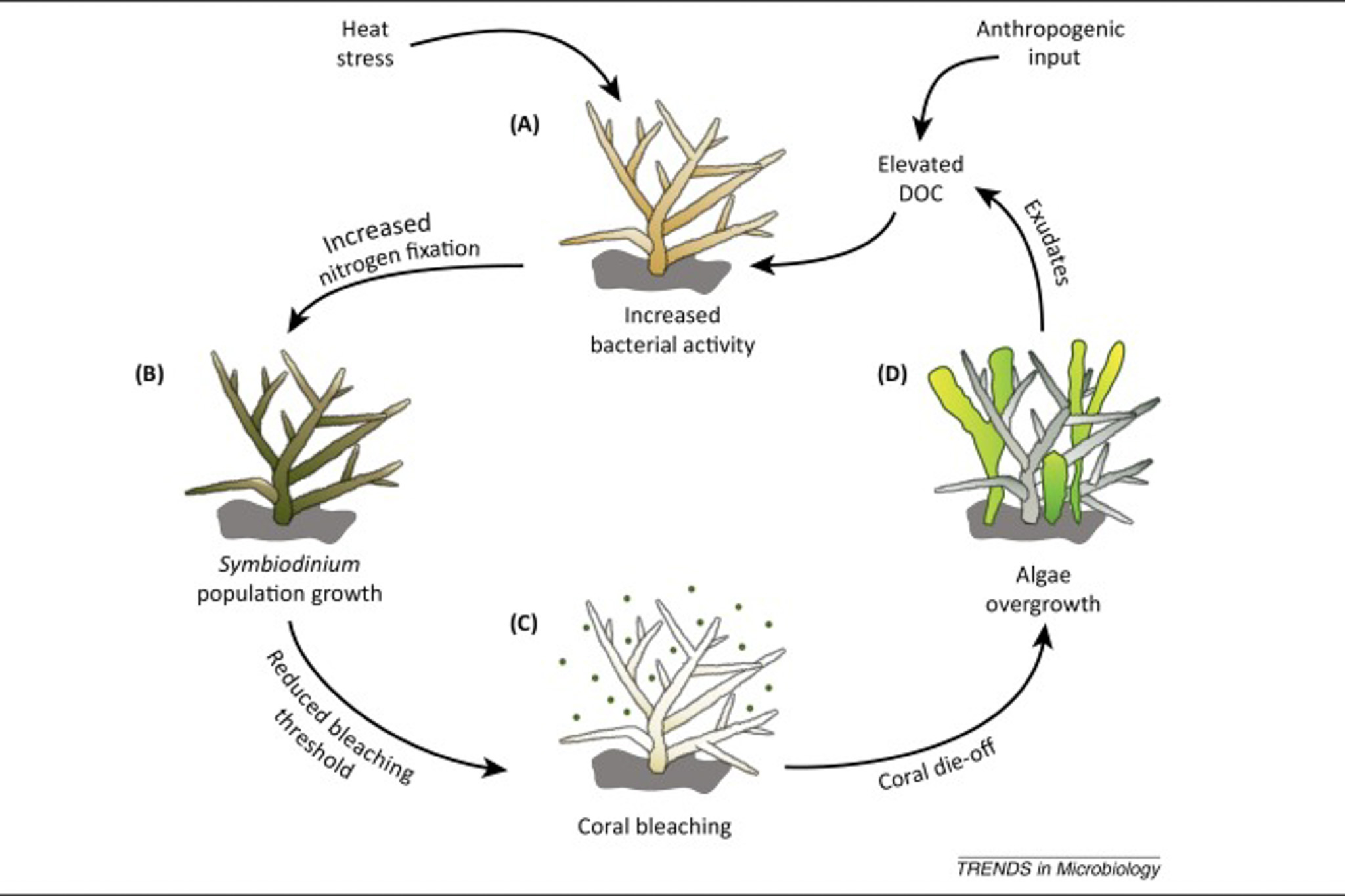
DDAM Model With Inclusion Of Nitrogen Fixation [6]
Overall, a multitude of studies seem to illustrate that excess inorganic nutrients (in particular nitrate) can cause complications with coral physiology making them much more susceptible to disease, bleaching and mortality.
Urea Doesn’t Have These Downfalls
Corals can obtain nitrogen from inorganic sources (N2, nitrate and ammonia) as well as organic sources (heterotrophic feeding, amino acids and urea). It’s been found that urea is found naturally on healthy reef environments and is one of the core sources of nitrogen for corals in oligotrophic (low nutrient environments).
I have spoken much about oligotrophic environments before. Without recapping things from the DOC article too much they are the state in which reefs are natrually found and corals have been determined to be healthiest. I am of the belief we should mimic these environments biochemically as best we can in reef tanks and our inability to do so is leading to many of the problems we face as aquarist.
In oligotrophic environments, urea concentrations can sometimes be 20 times higher than those of nitrate and ammonia. [7] Urea is utilized by most marine organisms and produced by larger vertebrates such as fish and marine mammals, seagrass beds, and many microbial processes. It’s a naturally occurring and important part of the nitrogen cycle on reefs and is likely one of the reasons Darwin’s Paradox is able to function.
Experiments conducted on Stylophora pistillata demonstrated the bacterial fraction of the coral uptakes close to 11% of available urea. During periods of light, there was a correlative increase in the uptake of urea. Most urea was found to be uptaken by the coral directly instead of the zooxanthella. This is likely because urea also plays a pivotal role in calcification.
“Campbell and Speeg (1969) suggested that urea could be involved in calcification by two mechanisms: i) the neutralization of protons formed during calcification with ammonia produced by urea hydrolysis; ii) the CO2 supply to the calcification, CO2 being a product of urea hydrolysis. Crossland and Barnes (1974) also found 14C in the skeleton of corals incubated in 14C urea.” [8]
A more recent experiment solidified the idea that urea is important to coral calcification. It even demonstrated that providing corals with urea and nickel led to faster growth rates. It was found that nickel is a cofactor for the urease enzyme (the enzyme which breaks down urea), thus supplying the coral with both, allowed it to effectively metabolize urea.
“Any process which increases the supply of Ca2+ and DIC to the calcification site, or the removal of H+ from this site, will likely enhance calcification. One stimulus can be urease, which catalyzes the hydrolysis of urea to produce inorganic carbon and ammonia (Krajewska, 2009). Ammonia can help neutralizing protons emitted during the calcification process, and thus help increasing the pH at the calcification site (Crossland and Barnes, 1974). Inorganic carbon produced by urease activity can stimulate calcification directly or indirectly, via increased photosynthesis, which also requires DIC (Allemand et al., 2004; Gattuso et al., 1999).” [9]

A comparison to the number of dead colonies after Nitrate and Urea addition [2]
Additionally, it was shown that urea is more easily absorbed than nitrate since it is a more energetically favorable process. It was ultimately found that Ni and urea enrichment increased calcification and photosynthetic rates, leading to faster growth. [9]
A recently submitted proteomics study, which has yet to be formally published, tracked the protein pathways associated with nitrogen metabolism. This study found that even the zooxanthellae benefit from urea.
“Physiology analysis showed that the symbiodiniacean species Cladocopium goreaui, which containing both URE and UAD, grew better under urea than ammonium conditions, as indicated by higher maximum specific growth rates. Furthermore, most genes of Symbiodiniaceae involved in urea utilization appeared to be stable under various conditions such as heat stress (HS), low light density, and nitrogen deficiency, wheras in ammonium and nitrate transporters were significantly regulated. These relatively stable molecular regulatory properties support sustained urea absorption by Symbiodiniaceae, as evidenced by an increase in ?15N2-urea absorption and the decreases in ?5N-NO3-, and ?15N-NH4+ from cultural environment to Symbiodiniaceae under HS conditions.” [10]
Overall, urea has been shown to be naturally occurring on healthy reefs, utilized by the bacteria, zooxanthellae and coral directly, act as a more favorable nitrogen source than ammonia and increase the growth and photosynthesis rates of coral.
Ammonia Isn’t All That Bad Either
Anyone that’s been dosing ammonia the last year or so knows – its the bees knees. I am not here to refute that. My own experience supports this and thankfully it seems the scientific literature does as well. As mentioned earlier corals need both inorganic and organic nitrogen DIN and DON. While urea serves as an excellent source of DON (and can even supply some ammonia when it’s broken down or dissociates) corals still need a good supply of DIN.
This section of the article will bring more evidence to the table on why ammonia dosing is likely a good thing and is certainly preferable to directly dosing nitrates.
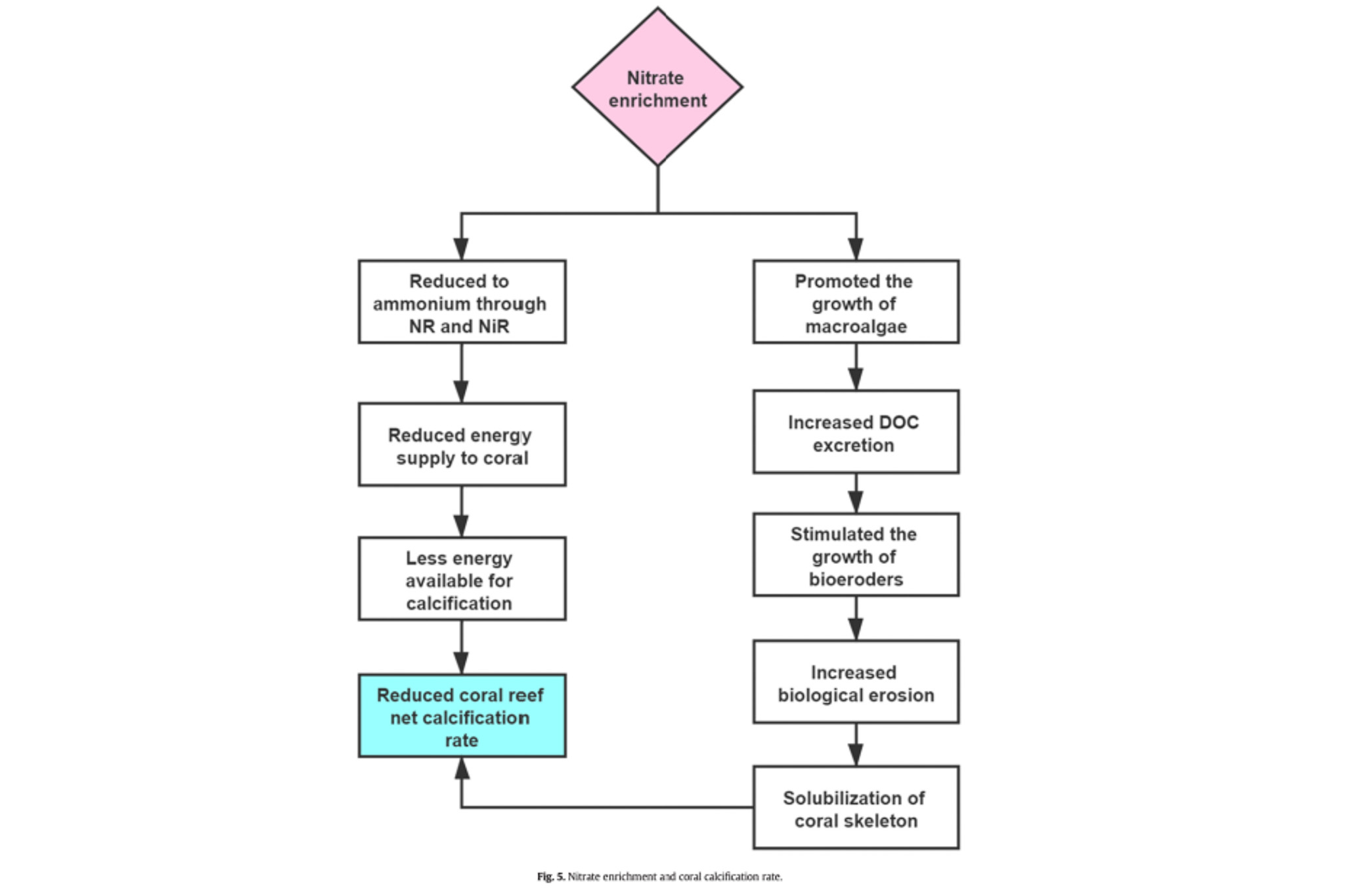
A recent study directly compares nitrate to ammonia addition. It found that even after stopping nitrate enrichment and thermal stress, the corals were unable to get back to ground zero and adequately repair themselves. High levels of free radicals persisted, and both zooxanthellae and chlorophyll levels stayed low. The most shocking result was that corals were no longer able to calcify, which heavily suggested they had a severe energy deficit. This creates long-term feedback where the coral struggles to heal and has to direct the little energy it has from low levels of zooxanthellae and chlorophyll into enzymes designed to combat free radicals. Thus, excess nitrate combined with external stress seems to be a long-term recipe for disaster that’s likely to keep corals down for the count.
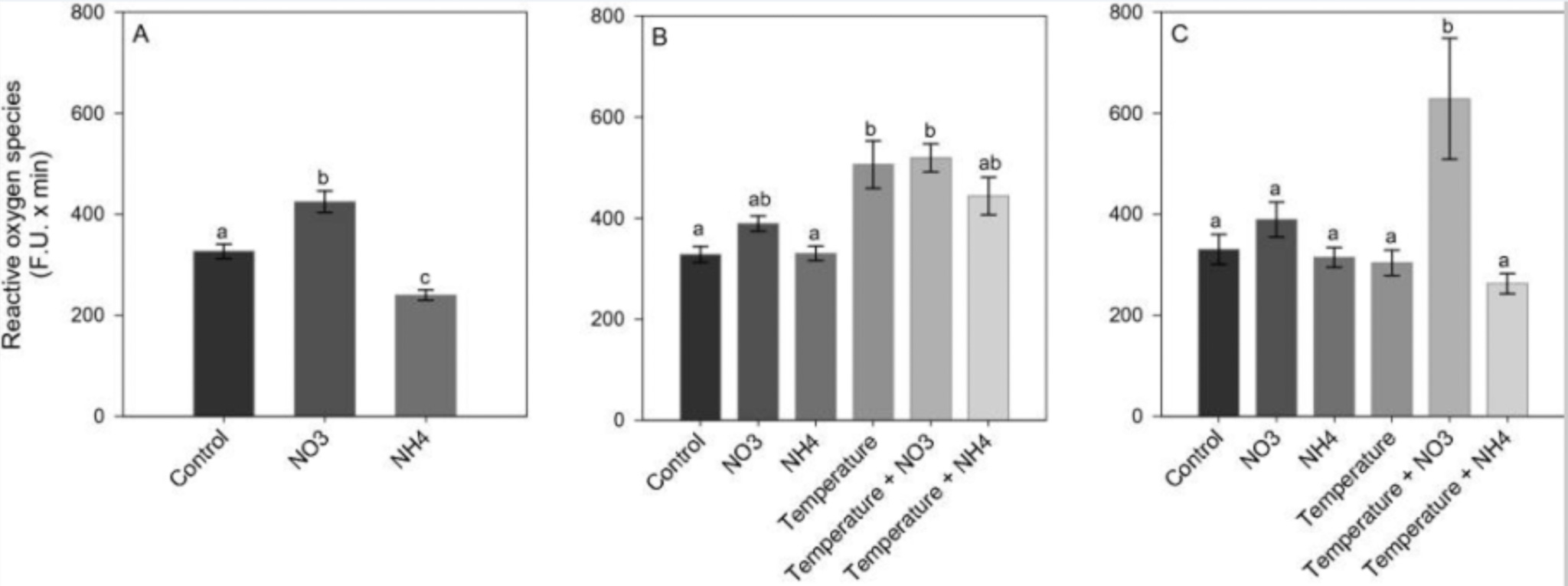
Comparison of reactive oxygen species between nitrate and ammonia additions [11]
In comparison ammonia enrichment aided the coral’s stress. It is theorized that ammonia assisted the coral in carbon acquisition and aided in the production of antioxidant compounds. Additionally, the study found after 5 weeks of ammonia enrichment there was a much high level of zooxanthella compared to control corals.
“Our results are in agreement with previous evidence showing that NH4+ enrichment can enlarge symbiont populations whilst also increasing thermal tolerance in corals and that NO3? enrichment can increase bleaching susceptibility without prior symbiont density enlargement16. Our findings also agree with the assertion of Morris et al.57, who suggested that the different impacts of ammonium and nitrate on coral thermal tolerance can be explained by their relative effects on the carbon metabolism and oxidative stress of the coral holobiont. We indeed showed that while nitrate enrichment increases oxidative stress in corals, ammonium enrichment tends to decrease it.” [11]
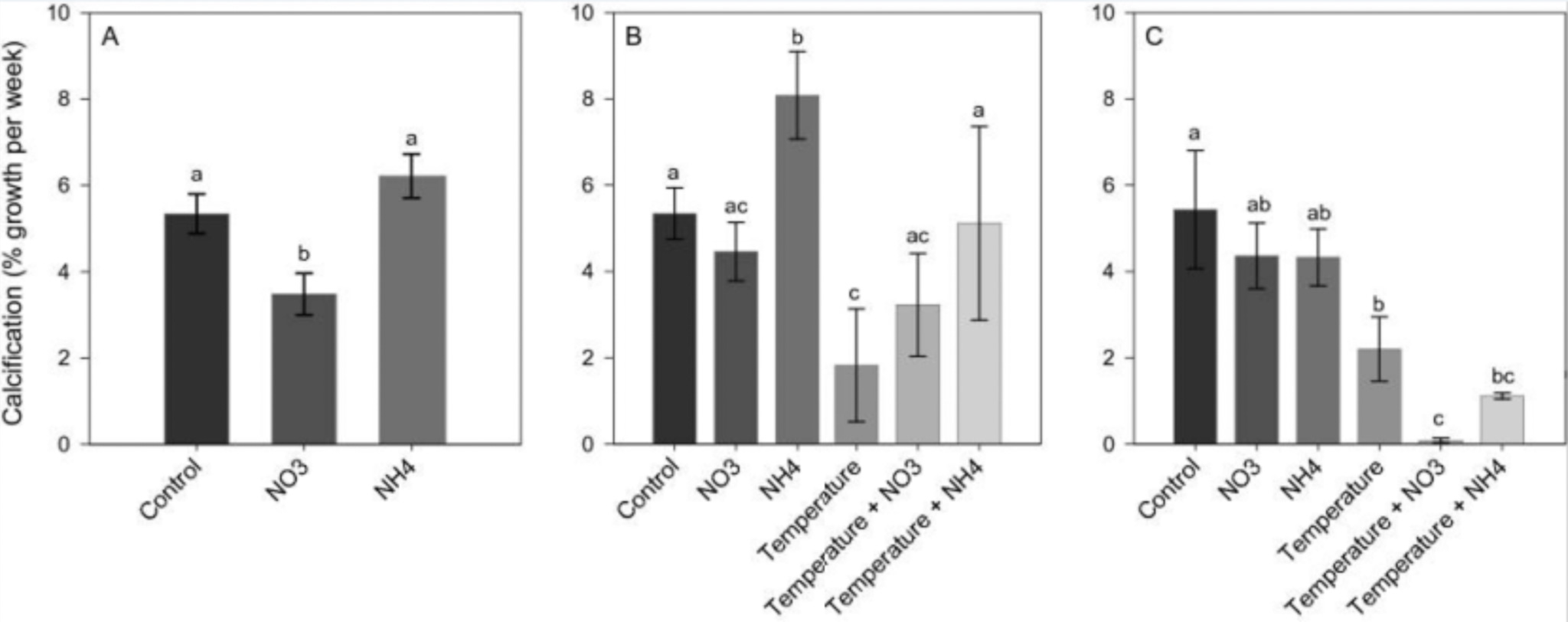
Comparison of calcification rate between nitrate and ammonia additions [11]
The Ultimate Nitrogen Supplement?
Thus since we’ve established corals would likely benefit from both ammonia and urea how should we dose it? At one point I read a study that suggested DON/urea makes up 1/4th of the nitrogen metabolism of corals. For the life of me cannot find this original study (I will continue to dig)
I think even if this is some sort of mind story or misremembrance the 1/4th ratio may be a good hubris to utilize. If we were to apply a 50/50 ratio of ammonia to urea we could overshoot things since urea dosing is relatively new and is not straight forward to monitor.
Thus, as a conservative approach, I suggest is mixing a ratio of 1:4 urea: ammonia. We will utilize the typical Randy Holmes-Farley ammonium bicarbonate recipe as our base for this.
Mixing Instructions
- Mix 20 g of ammonium bicarbonate per 1 liter of RODI water
- Add 5 g of urea to this mixture
I recommend adding ammonium bicarbonate first as it’s not as soluble in water as urea is. Thus by adding it first you can ensure it’s fully mixed. Be sure to wear PPE or use caution when mixing as the smell can be quite potent and you certainly don’t want to get any in an open cut or your eyes.
Dosing Instructions
If you’re already dosing ammonia you may employ the same approach I originally did. Decrease your dose by about 20-50% and then slowly increase it over time in order to stabilize nitrates/if you don’t see any issues.
If you’re not already dosing ammonia I recommend you slowlyyyyy transition from nitrate to this mixture – even dosing both at the same time. Slowly ramp down your nitrate addition and slowly ramp up the concoction dosage until your nitrates have stabilized and you’ve eliminated direct nitrate dosing.
For more help dialing in ammonia based dosing be sure to read Micheal Paletta’s original ammonia dosing Reef Builders article here.
Flow chart showing pathway between nitrate addition and bleaching [3]
Where Do I Purchase Urea?
You can purchase 1 lb of low grade here for $10.00 USD (I don’t recommend this)
You can purchase 1 lb of USP grade here for $40.00 USD (I recommend this)
You can purchase 500 grams of reagent grade here for $67.70 USD (For those with the budget of Andrew Sandler, I highly recommend this)
You can make urea at home using your own urine with this method for $0.00 (for those without the budget of Andrew Sandler – I highly recommend this)
How Do You Monitor Urea/DON Levels?
While overdosing urea is unlikely if you do so conservatively (since it is so widely metabolized), it wouldn’t hurt to periodically check your organic nitrogen levels. As of now, the only way on the market to do this is with a Triton N-DOC test, which can be purchased here. The N-DOC test also provides users with the most accurate reading of dissolved organic carbon (DOC) on the market, as it utilizes a total carbon analyzer instead of the spectrophotometric based methods (SAC/SAK) which many other testing companies have now adopted.
SAC/SAK methods rely on a correlation curve to give you results. How they work is you send in a water sample which is then put in a machine called a spectrophotometer. This machine passes UV light at a controllable wavelength through the sample. A detector on the other side of your sample then measures the interference your sample has with this beam of UV light. In this instance, the SAC test we are familiar with in the industry uses a beam of 254 nm UV light.
This beam of light can interact with many organic compounds – the compounds it “bumps” into most are often bulkier compounds which have rings (aromatic/cyclic) or multiple bonds (double, triple etc). While SAC254 readings do have a strong correlation with the overall DOC of aqueous systems the results still aren’t exact. That’s because not every carbon compound which makes up the DOC pool is detectable via the 254 nm wavelength.
Due to our lack of knowledge on the subject at this time it’s unknown if such an accurate reading is needed but nonetheless double checking your DOC and DON in a single test is likely worth it as a periodic redundancy measure.
Flow chart showing pathway between nitrate addition and slower growth [3]
For Those Who Use Or Produce Nitrate Additives
First, a point of clarification. I am in no way attacking you. My goal with this article is to attempt to generate conversation in the industry regarding the best method for coral care. We all do this for the animals, and they deserve our due diligence.
To people who use nitrate based products – please comment your thoughts on this article or in the comments on Facebook or Instagram.
To any company that is concerned with this article to I invite you to contact me directly at [email protected]. I also invite any company that may have qualms with this approach to publicly come on the Reef Therapy podcast and have a discussion with me about it.
Conservation, civil discourse, and debate are how we, as a community and industry, are able to move forward, and those are all things I wish to facilitate through Reef Builders.
To anyone that may work for a company reading this paragraph I encourage you to invest in specific R&D to help answer the question of “what happens when someone adds this to their tank?”. Companies answering these questions themselves with their own data and experiments could accelerate this industry forward a great deal.
It has yet to be 100 years since coral was first kept in a glass box. We likely only have 100 more until most coral is kept in glass boxes. I hope through continued research and conversation we can all work to build reefs together.
Sources:
[1] Donovan, M. K., Adam, T. C., Shantz, A. A., Speare, K. E., Munsterman, K. S., Rice, M. M., Schmitt, R. J., Holbrook, S. J., & Burkepile, D. E. (2020). Nitrogen pollution interacts with heat stress to increase coral bleaching across the seascape. Proceedings of the National Academy of Sciences, 117(10), 5351–5357. https://doi.org/10.1073/pnas.1915395117
[2] Burkepile, D. E., Shantz, A. A., Adam, T. C., Munsterman, K. S., Speare, K. E., Ladd, M. C., Rice, M. M., Ezzat, L., McIlroy, S., Wong, J. C. Y., Baker, D. M., Brooks, A. J., Schmitt, R. J., & Holbrook, S. J. (2019). Nitrogen Identity Drives Differential Impacts of Nutrients on Coral Bleaching and Mortality. Ecosystems, 23(4), 798–811. https://doi.org/10.1007/s10021-019-00433-2
[3] Zhao, H., Yuan, M., Strokal, M., Wu, H. C., Liu, X., Murk, A., Kroeze, C., & Osinga, R. (2021). Impacts of nitrogen pollution on corals in the context of global climate change and potential strategies to conserve coral reefs. Science of the Total Environment, 774, 145017. https://doi.org/10.1016/j.scitotenv.2021.145017
[?4] Yang, H., Yuan, D., Zhou, Z., & Zhao, H. (2025). Nitrate enrichment exacerbates microbiome and metabolism disturbances of the coral holobiont under heat stress. Marine Environmental Research, 208, 107098. https://doi.org/10.1016/j.marenvres.2025.107098
?[5] Vega Thurber, R. L., Burkepile, D. E., Fuchs, C., Shantz, A. A., McMinds, R., & Zaneveld, J. R. (2013). Chronic nutrient enrichment increases prevalence and severity of coral disease and bleaching. Global Change Biology, 20(2), 544–554. https://doi.org/10.1111/gcb.12450
?[6] Rädecker, N., Pogoreutz, C., Voolstra, C. R., Wiedenmann, J., & Wild, C. (2015). Nitrogen Cycling in Corals: the Key to Understanding Holobiont Functioning? Trends in Microbiology, 23(8), 490–497. https://doi.org/10.1016/j.tim.2015.03.008
[7] Crandall, J. B., & Teece, M. A. (2011, October 30). Urea is a dynamic pool of bioavailable nitrogen in coral reefs – coral reefs. SpringerLink. https://link.springer.com/article/10.1007/s00338-011-0836-1
[8] Grover, R., Maguer, J.-F., Allemand, D., & Ferrier-Pagès, C. (2006). Urea uptake by the scleractinian coral Stylophora pistillata. Journal of Experimental Marine Biology and Ecology, 332(2), 216–225. https://doi.org/10.1016/j.jembe.2005.11.020
[9] Biscéré, T., Ferrier-Pagès, C., Grover, R., Gilbert, A., Rottier, C., Wright, A., Payri, C., & Houlbrèque, F. (2018). Enhancement of coral calcification via the interplay of nickel and urease. Aquatic Toxicology, 200, 247–256. https://doi.org/10.1016/j.aquatox.2018.05.013
[10] EMBL-EBI. (2025). PRIDE – PRoteomics IDEntifications Database. Ebi.ac.uk. https://www.ebi.ac.uk/pride/archive/projects/PXD056312
[11] Fernandes de Barros Marangoni, L., Ferrier-Pagès, C., Rottier, C., Bianchini, A., & Grover, R. (2020). Unravelling the different causes of nitrate and ammonium effects on coral bleaching. Scientific Reports, 10(1), 11975. https://doi.org/10.1038/s41598-020-68916-0




Thanks for a good and firmly detailed article. Your knowledge sharing are really appreciated.
How does excretion of ammonia and urea from fish goes into the equation ?
I’ve been dosing ammonia for like 4 months, but stopped as I did not see any changes on the corals, and wondering if the addition of urea might do a difference.
I have a 180G well stocked tank with primarily sps corals and 35 well feed fish, so I expect they expels a lot of nitrogen in different forms. But no idea if the amount of nitrogen produced by the fish even come close to the need of nitrogen from the corals
Fascinating read! The exploration of urea dosing as a nitrogen source challenges traditional reefkeeping paradigms, particularly the long-held aversion to organic nitrogen. The idea that controlled urea supplementation could mimic natural reef conditions—where corals likely absorb organic nitrogen compounds—is compelling. The comparison to terrestrial plant fertilization (NH4+ vs. urea) adds an interesting perspective, though I’d love to see more empirical data on coral uptake efficiency and potential risks like bacterial blooms or unintended symbiont shifts.
The emphasis on balance (e.g., maintaining phosphate ratios) resonates with recent trends toward holistic nutrient management, moving beyond the ‘zero-nitrogen’ mindset. Could this approach also benefit non-photosynthetic corals or tanks with heavy fish bioloads? Kudos for diving into the ‘Don Din rabbit hole’—this feels like the start of a much broader conversation!